TEPEZZA LAWSUIT FOR HEARING LOSS
Tepezza Lawsuit Update August 2023

As of August 2023, the new Tepezza class action MDL has consolidated all existing cases, with Judge Tomas Durkin presiding over the process. The first status conference has been held, and litigation continues to proceed with a current volume of 42 cases. Our attorneys expect this number to increase substantially over the coming months.
If you have used Tepezza to treat thyroid eye disease and subsequently suffered from tinnitus or hearing loss, you may have a viable product liability lawsuit. The nationally-recognized defective drug attorneys at Reich & Binstock are currently investigating Tepezza hearing loss lawsuits. To see if you qualify to file a Tepezza hearing loss lawsuit, please call our office at 713-622-7271 to schedule your free consultation.
What Is Tepezza?
Tepezza (teprotumumab-trbw) is the only FDA-approved medication for treating Thyroid Eye Disease (TED), also called Graves’ orbitopathy. It is an infusion drug that is administered intravenously over a 90-minute period for each treatment session. Generally, patients receive these infusions once every three weeks, totaling 8 infusions over the course of around 5 months.
Tepezza Side Effects
According to the Tepezza website, the most common Tepezza side effects and the percentages of patients who experience them are as follows.
- Nausea – 17%
- Hair loss (Alopecia) – 13%
- Muscle spasms – 25%
- Fatigue – 12%
- Diarrhea – 12%
- High blood sugar – 10%
- Changes in taste (Dysgeusia) – 8%
- Hearing problems – 10%
- Dry skin – 8%
- Headaches – 8%
- Weight loss – 6%
- Nail disorder – 6%
- Changes in menstruation – 23% of menstruating women
Tepezza can also cause certain infusion reactions either during treatment or within 24 hours of treatment. Common symptoms of Tepezza infusion reactions include rapid heartbeat, headache, difficulty breathing, muscle pain, high blood pressure, feeling hot, and redness in the face. Additionally, Tepezza may worsen one’s symptoms if the patient has Crohn’s disease, ulcerative colitis, or inflammatory bowel disease (IBD).
Is Tepezza FDA Approved?
Yes. Tepezza was approved in January 2020 to treat thyroid eye disease (TED). It is approved for specific use in patients who have had the disease for 9 months or less and who have seen higher activity levels from the disease. Tepezza is currently the only drug approved by the FDA to treat thyroid eye disease.
What Is Tepezza Used to Treat?
Tepezza is used to treat thyroid eye disease, eye bulging, eye pain, and double vision. It is a 24-week course of 8 IV infusions, with one infusion occurring once every three weeks.
What Is Thyroid Eye Disease?
Thyroid eye disease (TED) is a rare autoimmune disease that causes the fatty tissues and muscles behind the eye to become inflamed. The inflammation pushes the eyes forward, which leads to eye bulging, eye redness, and swelling around the eyes.
TED has two distinct phases – the active (inflammatory) phase and the stable phase. During the active phase, inflammation can last for several months or up to three years. The active phase occurs once the inflammation subsides.
The disease occurs more often in women than in men, mostly because this is also the case for hyperthyroidism. TED is related to hyperthyroidism and Graves’ disease, which both occur more commonly in women.
How Rare Is Thyroid Eye Disease (TED)?
According to the American Academy of Ophthalmology, thyroid eye disease has an incidence rate of around 19 in 100,000 per year (0.00019%). Around 16 per 100,000 women have this disease, while only 2.9 per 100,000 men have it. Thus, although TED is a rare disease, it occurs much more often in women than in men.
How Does Tepezza Work?
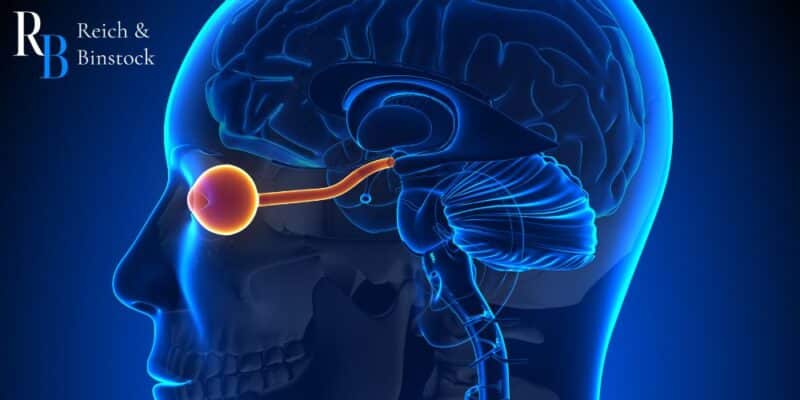
Tepezza targets not just the symptoms of TED, but also the source. It is a biologic inhibitor of insulin-like growth factor-1 receptor, also called IGF-1R. Once administered, Tepezza works to decrease inflammation, prevent the expansion of tissue behind the eye, and prevent fat tissue and muscle remodeling behind the eye.
Within the tissues behind the eyes are cells with surface receptors that function like light switches. When someone has TED, their immune system attacks these receptors and “turns on the light switch,” which then causes inflammation, eye bulging, eye pain, and double vision.
What Injuries Can Tepezza Cause?
According to ongoing Tepezza lawsuits, the drug has been linked to very serious injuries, including the following.
- Tinnitus (ear ringing)
- Partial hearing loss
- Permanent hearing loss
- Sensorineural hearing loss
- Increased sound sensitivity
- Ear plugging sensation
- Patulous eustachian tube dysfunction
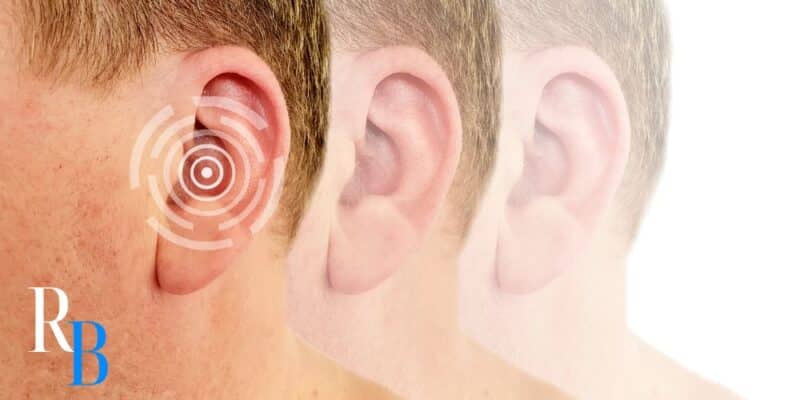
Can Tepezza Cause Hearing Loss?
Yes. Certain clinical studies have shown that Tepezza can cause partial or permanent hearing loss, or even total hearing loss. Other hearing symptoms, such as tinnitus and muffled hearing, are also possible.
How Long Does Hearing Loss from Tepezza Last?
It depends. If patients regularly monitor themselves for hearing loss or tinnitus, they may be able to catch the side effect before the drug causes irreversible damage.
Is Tepezza Hearing Loss Reversible?
In many cases, no. Tepezza can cause permanent hearing damage that is not reversible if patients do not detect the damage early enough, or if they experience ototoxicity caused by the drug. Ototoxicity is a side effect of the medication that damages the auditory nerve, which may cause temporary or permanent hearing loss.
Is There a Tepezza Class Action Lawsuit?
Technically, no. The Tepezza cases that have been filed have been consolidated into an MDL (multidistrict litigation) case. It is very common for class action MDLs to start slow and gain more traction over time. However, we expect this to be on the smaller side as far as MDLs go, which is good news for claimants. When these cases have fewer plaintiffs, this often means higher settlement amounts.
What Are the Tepezza Lawsuits About?
Tepezza lawsuits claim that the manufacturer of the drug, Horizon Therapeutics, PLC, failed to warn patients and doctors about the potential permanence of the hearing loss side effect. The full range of allegations against Horizon includes the following.
Failure to Warn and Inadequate Instructions
Horizon Therapeutics, PLC listed hearing loss as a potential side effect of Tepezza infusions, but did not mention potentially permanent hearing loss as a side effect. Their failure to warn of this very serious side effect prevented patients and physicians from making informed decisions about their TED treatment.
Tepezza hearing loss claims allege that Horizon knew of the possibility of permanent hearing loss as a side effect. Still, they did not include that information in their product’s packaging information. They also failed to inform physicians of the necessity of regular hearing tests over the duration of the treatment. If they had instructed doctors to monitor their patients’ hearing, this could have allowed for early detection and minimization of hearing damage.
Insufficient Safety Testing
Even though the drug received FDA approval, Tepezza hearing loss lawsuits allege that Horizon did not conduct adequate safety tests before seeking FDA approval. In March 2021, more than a year after the FDA approved the product, the Endocrine Society published a study that reported hearing problems in 65% of patients who received Tepezza. This figure is much higher than Horizon’s claim that only 10% of patients treated with Tepezza developed hearing impairment.
Overly Aggressive Marketing
The European Group on Graves’ Orbitopathy recommended Tepezza as a second-line treatment, which is a treatment that is initiated after the first-line treatment fails in some way or stops working. However, Horizon marketed Tepezza as a first-line treatment for TED.
Horizon marketed its product to millions of patients and physicians, even though the European Group on Graves’ Orbitopathy only recommends the drug to a small percentage of patients.
Failing to Update the Product’s Label
After multiple clinical studies and patients reported a strong connection between Tepezza and permanent hearing loss, Horizon failed to update their product’s labeling to reflect this information.
How to File a Tepezza Lawsuit
If you have developed hearing-related problems after taking Tepezza for TED, you may have the basis for a Tepezza hearing damage lawsuit. Before formally filing your claim, we strongly recommend speaking with a product liability lawyer with experience in defective drug cases. A skilled Tepezza lawyer will help you navigate the claims process as smoothly as possible, as well as assess the potential damages you may be entitled to.
What Evidence Do Victims Need for a Tepezza Lawsuit?
After you speak with an attorney about a Tepezza hearing loss lawsuit, gathering evidence should be a priority. It is extremely important to gather sufficient evidence to prove your claim. To have a successful claim, you will need high-quality evidence that shows the connection between your use of Tepezza and your hearing loss.
Examples of important forms of evidence include the following.
- Medical records and documentation
- Medical expenses and receipts
- Your Tepezza prescription
- Your personal testimony on your experience with side effects from the thyroid eye disease drug
- Evidence of costs related to your hearing loss symptoms, such as receipts for hearing aids
Who Is Liable for Tepezza Lawsuit Damages?
Horizon Therapeutics, PLC is the manufacturer and distributor of Tepezza. They were acquired in a merger by Amgen, Inc. Horizon is already a potentially liable party, but Amgen may also be held liable if the merger is approved for business purposes in the United States. Amgen could stop the sale of Tepezza, but they could still face liability regardless.
The doctrine of strict liability in product liability law means that any party in the chain of distribution could be held liable for Tepezza hearing loss lawsuits if a consumer injured by their product can prove one of the following elements.
They must prove that the product:
- Was inherently unsafe
- Did not have adequate labels, instructions, or warnings
- Had a defect during the manufacturing process
The doctrine of strict liability can even apply in cases where a manufacturer was not negligent. The injured consumer need only show that the manufacturer knew or should have known of the possibility of permanent hearing damage, and failed to warn the public of that fact.
Who Is Eligible to File a Tepezza Lawsuit?
To be eligible to file a Tepezza lawsuit for hearing loss, you must have received Tepezza infusions. You must also have suffered one of the following hearing problems in either one or both ears.
- Tinnitus
- Hearing impairment
- Muffled hearing
- Complete deafness
By meeting with one of our Tepezza lawyers, you can determine whether or not you qualify to file a Tepezza hearing loss lawsuit. We can evaluate the facts of your case during your free consultation and advise you on the best course of action.
What Damages Are Available in Tepezza Lawsuits?
If you have suffered hearing loss as a result of Tepezza infusions, the possible forms of economic and non-economic compensation in Tepezza lawsuits include the following.
- Current and future medical expenses
- Expenses for hearing tests and hearing aids
- Current and future lost wages
- Loss of earning capacity
- Pain and suffering
- Loss of one of the senses
Other forms of compensation may be available, and one of our attorneys will inform you of all possible options when you file a Tepezza lawsuit.
Potential Tepezza Settlement Amounts
It is extremely difficult to estimate the potential value of Tepezza lawsuits without carefully examining the facts of each individual case. Even if you see an “average” number online for Tepezza settlement amounts, this number could be highly inaccurate for your case. The bigger the negative impact that Tepezza has on a patient’s life, the higher the value of their claim is likely to be.
How Long Do I Have to File a Tepezza Claim?
Each state has its own statute of limitations regarding product liability cases. The best way to know how much time you have to file is to speak with a Tepezza attorney as soon as possible. Although our law firm handles defective drug cases from across the nation, we are based in Texas. In Texas, claimants generally have two years from the date of their injury to initiate a Tepezza lawsuit.
To ensure that you do not miss your filing deadline, contact a skilled personal injury lawyer with Reich & Binstock as soon as possible. We will evaluate the facts of your case and advise you of whether you have a valid claim. Working with an attorney will ensure that you do not miss your filing deadline or your right to fight for compensation.
Why Choose Reich & Binstock for Your Tepezza Claim?
Drug manufacturers are very large companies that often have teams of lawyers on their side. For this reason, it’s important to work with attorneys with extensive experience handling dangerous drug claims. At Reich & Binstock, our defective drug lawyers are nationally-recognized for our compassionate support and aggressive representation of clients.
With decades of litigation experience on our side, we have what it takes to face even the biggest pharmaceutical companies and achieve positive outcomes for our clients. Our attorneys have secured significant recoveries for our clients, and we have a long history of achieving extraordinary settlements and verdicts.
If you are a Tepezza patient who has suffered permanent hearing loss or other auditory issues, our law firm is here for you.
Contact Reich & Binstock for Help Filing a Tepezza Claim
If you or a loved one suffered permanent hearing problems after taking Tepezza, you deserve compensation for your injuries. To find out if you are eligible for a Tepezza lawsuit, contact the law firm of Reich & Binstock. Call our office today at 713-622-7271 to schedule your free consultation with us. We operate on a contingency fee basis, meaning we don’t accept a cent unless we secure a recovery on your behalf.
Our law firm is based in Houston, TX, but we are currently accepting and investigating nationwide Tepezza hearing damage lawsuits.
There is never a fee unless we recover on your behalf.